Understanding the relevance of chemical equations


Introduction
Atoms of various elements combine to form molecules and compounds. In previous articles, we have discussed various types of chemical combinations such as covalent and electrovalent bonding. Reporting such chemical reactions on paper usually involves writing the symbols, formulae and number of reacting atoms, plus the compounds or molecules formed.
Chemical equations usually show the reactants on one side and products formed on the other side. Reactants are written on the left side of the equation while products are written on the right side of the equation. After writing a chemical reaction, the number of atoms on either sides of the equation should be the same. Remember the law of conservation of matter. The law shows that while matter can be converted from one form to another, it can neither be created nor destroyed. So every chemical equation must be balanced.
So after writing a chemical reaction, the number of atoms on either sides of the equation must be the same. If the atoms on one side are not the same in number as those on the other side, then coefficients have to be added in front of the reactants and products to balance the equation.

Understanding the components of a chemical reaction.
We will use a sample now and try to explain the various parts or components of a chemical reaction.
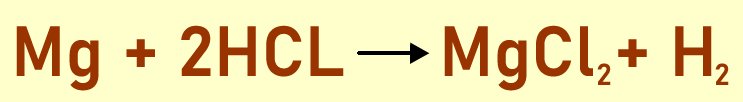
This is a sample chemical reaction and we can use it to understand the various components involved when writing a chemical reaction:
The reactants: The atoms combining to form a new molecule of compound are known as the reactants. They are usually written at the left side of the equation. In above example, the reactants are magnesium and hydrogen chloride. The 2 moles of HCL combines with Mg and both of them are the reactants in the above equation.
The arrow: There is always an arrow after writing the reactants. The arrow indicates the compounds or molecules formed after the reaction was completed.
The products: After a chemical reaction, new molecules or compounds are formed. These are written at the right hand side of the equation just after the arrow. So for the above example, a mole of Magnesium chloride is formed one mole of hydrogen molecules.
Once a chemical reaction like the one above is complete, it is important to check the number of atoms at the beginning of the reaction and the number at the end to see if they match. The law of conservation of matter must be obeyed. If there are discrepancies between the number of reacting atoms and those of the products, the chemical equation must be balanced. To do this one, coefficients are added in from of the atoms such that there is an equal number of reactants and products.

Why write chemical equations?
Chemical reactions are so important in modern chemistry because they provide valuable information to the scientist, especially when he is not there to perform an experiment in person.
We will use an example to see the value of chemical equations in modern chemistry. Consider the following equations:
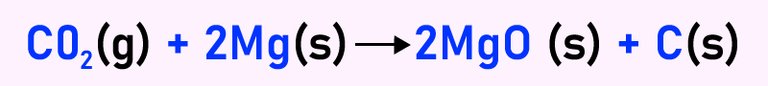
We can gather the following valuable information from a checmical equation like the one above:
1. The reactants: Looking at a chemical equation, we can easily spot the reactants at the left side of the equation. The above reaction shows the decomposition of carbon (iv) oxide by the heat produced from a burning magnesium. So the reactants are Magnesium and Carbon (iv) oxide.
2. Elements in the reaction: In the above example, we can easily see the individual elements that make up the reactants. They include Carbon (C), Magnesium(Mg), Oxygen(O).
3. Products: The products are found on the right side after the arrow. In this case, we can see that after the decomposition, carbon and Magnesium oxide are the products.
4. Reversible/Irreversible reaction: We can know whether a chemical reaction s reversible or not by looking at the equation. If there is one arrow pointing towards the right side, it is an irreversible reaction. It simply means that we cannot form the reactants from the product. However, if there are two arrows one pointing to the left and another pointing to the right, the reaction is reversible. That simply means that the reactants could be formed from the products.
5. State of matter: From a chemical equation, one should easily deduce the states of matter in the reactants and products. These are usually indicated with a letter inside a bracket. For example, Carbon (iv) oxide is a gas, so it was denoted with (g). Magnesium is a solid (s), Carbon is a solid (s), and Magnesium oxide is also a solid denoted with the initials (s).

Conclusion
Chemical equations are really important for every reaction because it helps scientist to understand a lot about the reaction and products. It also is a great way to report or document the results obtained at the end of an experiment.
In the next class, we will see how to balance chemical equations!!!!
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider delegating to the @stemsocial account (85% of the curation rewards are returned).
Thanks for including @stemsocial as a beneficiary, which gives you stronger support.